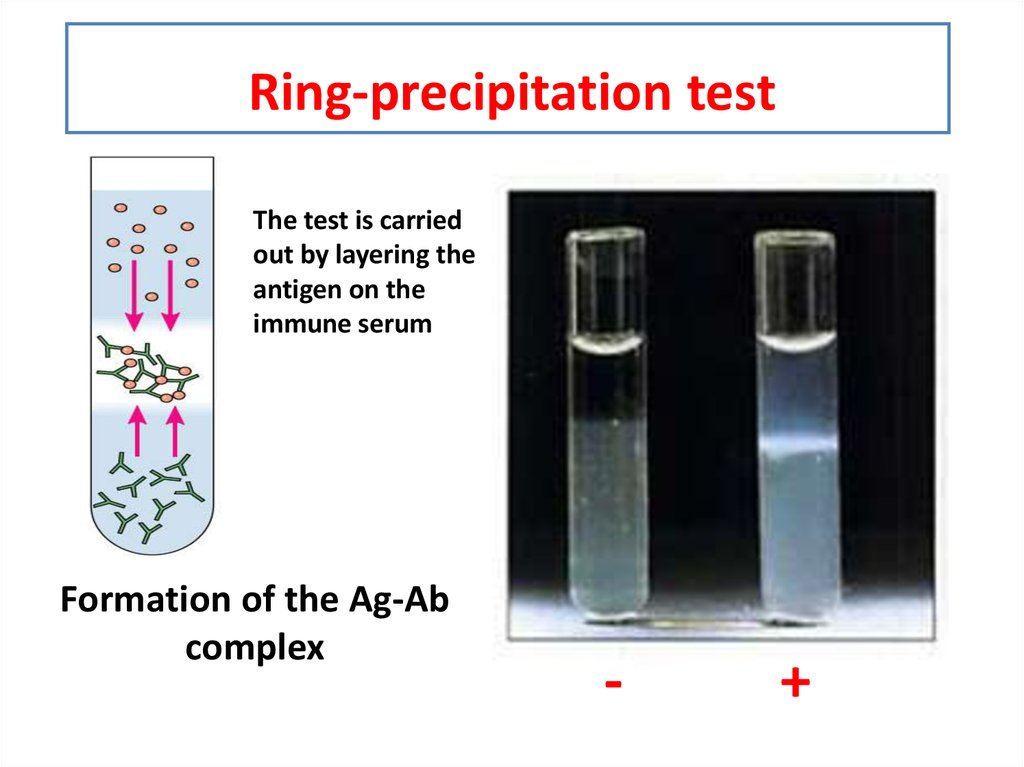 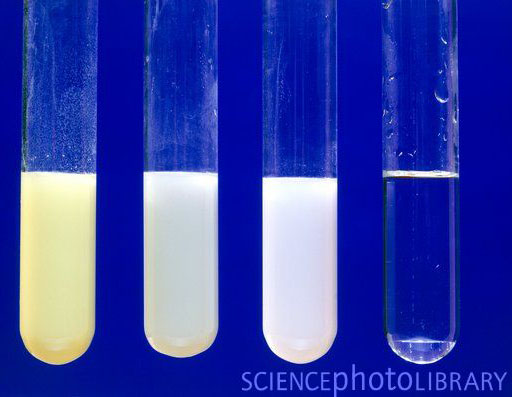
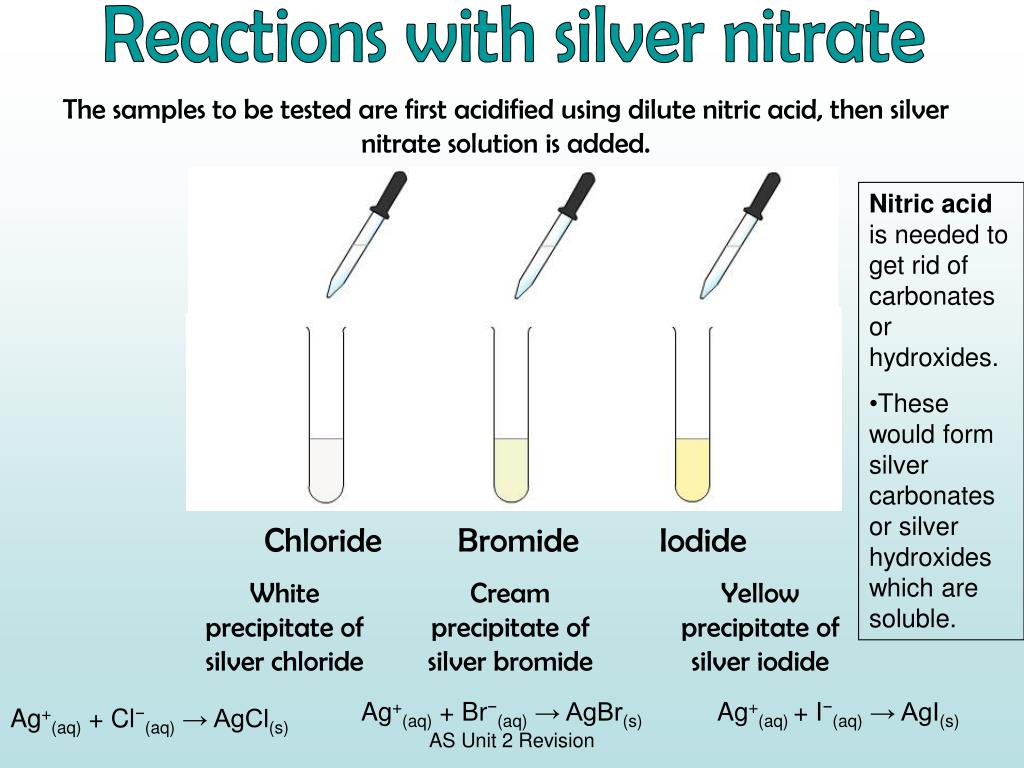
* Carry out simple precipitate tests to identify halide, sulfate and carbonate ions **Lesson 3 - Identifying negative non-metal ions * Describe the problems and limitations of using flame tests and precipitate tests to identify positive metal ions * Describe how to carry out a flame test and a precipitate test, including the names of any important reactants * Carry out simple precipitate tests to identify positive metal ions * Carry out simple flame tests to identify positive metal ions **Lesson 1-2: Identifying positive metal ions (flame tests and precipitate tests) **Includes 4 LESSONS worth of teaching materials:** Risk assessment/order form - containing up to date CLEAPPS guidance as of Oct 2023.Ī bundle containing everything you need to teach the GCSE Triple Chemistry content for AQA Chemical Analysis.Student worksheet answers (PDF and editable word versions).Student worksheet - including practical instructions, tables, and practical quesitons (PDF and editable word versions).Lesson powerpoint - including starter activity, practical instructions, tables, challenge task, multiple choice quiz plenary, and full answers.Write balanced symbol and ionic equations for the reactions taking place in precipitation reactions.Describe how to carry out precipitate tests to test for halide, sulfate and carbonate ions, including the names of any important reactants.Carry out simple precipitate tests to identify halide, sulfate and carbonate ions.This resource is designed for the AQA Triple Chemistry required practical from “Chemical Analysis”, and is relevant to higher and foundation students. In these reactions, bond strength is the main factor deciding the relative rates of reaction.A lesson pack covering negative non-metal ion tests (halide, sulfate and carbonate). However, the fastest reaction is with an iodoalkane. The electronegativity difference between carbon and iodine is negligible. 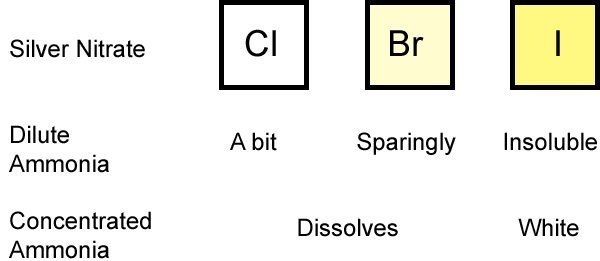
That means that there will be more attraction between a lone pair on the water and a carbon atom attached to a chlorine atom than if it was attached to an iodine atom. The slight positive charge on the carbon will be larger if it is attached to a chlorine atom than to an iodine atom. It is tempting to think that the reaction will be faster if the electronegativity difference is greater. It is slightly positive because most of the halogens are more electronegative than carbon, and so pull electrons away from the carbon. If you have looked at the mechanisms for these reactions, you will know that a lone pair on a water molecule attacks the slightly positive carbon atom attached to the halogen. In order for a halide ion to be produced, the carbon-halogen bond has to be broken. The carbon-iodine bond is the weakest and the carbon-chlorine the strongest of the three bonds. The order of reactivity reflects the strengths of the carbon-halogen bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
